Gary Taubes vs Stephan Guyenet on Joe Rogan Podcast
If you haven't yet seen the Gary Taubes vs Stephan Guyenet showdown on Joe Rogan's podcast last week, none of this will make any sense. I’d like to give my thoughts on the Stephen Guyenet vs Gary Taubes debate on the Joe Rogan podcast. I know everyone is tired of this discussion, so hopefully, I bring something new that you haven’t heard yet. Here goes. Initially, I expected Stephan to wipe the floor with Taubes. I did. I understand the importance of calories, and I think the carb-insulin...


If you haven't yet seen the Gary Taubes vs Stephan Guyenet showdown on Joe Rogan's podcast last week, none of this will make any sense. I’d like to give my thoughts on the Stephen Guyenet vs Gary Taubes debate on the Joe Rogan podcast. I know everyone is tired of this discussion, so hopefully, I bring something new that you haven’t heard yet. Here goes. Initially, I expected Stephan to wipe the floor with Taubes. I did. I understand the importance of calories, and I think the carb-insulin hypothesis is terrible. To be fair, my bias here was clearly leaning for Stephan. But as I watched the debate, I noticed a lot of things that I found to be fairly bothersome. In an ideal, goal-oriented world, this argument would not have been nearly as terse or personal as it had gotten. No one would hold themselves in such high esteem that they wouldn’t take counter-points even if anecdotal at face value for the sake of teasing out the nuance in such an important discussion. No one would fail to acknowledge that at some point in time that they actually agreed that the insulin hypothesis did hold some plausibility. No one would fail to see that both sides bring something of equal importance to the table. No one would dismiss the other off-hand. In an ideal world, perhaps we would recognize that these two very intelligent individuals are unfortunately committing the logical fallacy of composition. One, that what is true of the whole is applicable and reflective of the individual. The other, that what is true of the individual is a reflection of the whole. If CICO is the overwhelming driver of obesity for the whole, it does not inherently mean it is applicable on an individual level. If obesity in an individual is driven by carbohydrate intake and insulin it does not inherently mean this is the driver of obesity for the whole. If the goal is to discover what the multitude of causes of obesity is for the sake of progress, these two would have seen each other for what they are and worked as a team to look for answers and directions. What Stephan failed to recognize is that Gary is poking holes in his theory by using anecdotes and outliers. Folks, this is indicative that there remains more to be seen in the individual and outliers. What Gary failed to recognize is that Stephan has a plethora of evidence to support what is happening to the group, and therefore he needed to recognize that his theories did not hold up to scrutiny for the whole. But alas, the dichotomania persists. If you went in rooting for Taubes, you likely came out rooting for Taubes. If you went in rooting for Stephan, you may have ignored his behavior and likely came out rooting for Stephan. And with the dichotomania, we failed to recognize the two very important perspectives that each brought to the table. I came out irritated, and by the time I was reading everyone else’s reviews I was thinking “Wow, how good we are as humans to squander knowledge of others because we don’t recognize the value of people who think differently from us.” We didn’t see it as them being both right and both wrong. But they were.
“Taubes is arguing with stories.” Even if his conclusions and theories are dubious, way to expertly diminish the fact that his gift is in poking holes and pointing out that there is something wrong with the current paradigm on the causes of obesity. “Stephan was an arrogant asshole.” Who gives a shit.
Stephan has so much information he could be dressed as a unicorn wearing hot pink lipstick and making horse noises, and you’d be an idiot not to move past that fact and consider everything he says carefully. If you have the best brain surgeon in the world operating on your brain tumor but his bedside manner sucks, are you going to find someone else to perform the surgery who is “nice?”
But to avoid my own hypocrisy in writing this post, I’m going to be goal-oriented here as I wish they had been.
How Taubes and Guyenet Were Both Right and Wrong in My Estimation
First, it is clear to me that both Taubes and Stephan were both right and wrong. Second, it is clear to me that we are missing some big piece which makes them both right. Third, I’ve said this a million times since 2012, everyone needs to take a seat and chill the f out until we work out the kinks and nuances of the gut microbiome. As far as the gut microbiome is concerned, thus far we understand that: • Microbiota produce metabolites and microbial products like lipopolysaccharides (LPS) • Lipopolysaccharides (or endotoxins) are part of the outer membrane of Gram-negative bacteria. • In gut dysbiosis featuring more gram-negative bacteria, more LPS is presented. • LPS stimulates the production of inflammatory cytokines within the central nervous system, especially the pineal gland that produces melatonin (more on that in a bit) • LPS can modulate appetite, energy uptake, storage, and energy expenditure. • LPS shifts post-prandial plasma ghrelin to *fasted levels.* (in case you would like a mechanism for how gut dysbiosis itself can lead to overeating) • LPS promotes leptin resistance • LPS down-regulates the expression of neuropeptide proglucagon • LPS inhibits expression of gut-secreted GLP-1 and PYY (so increases food intake, and anxiety and depression-related behavior) (Sidebar: while some specialists see the link between depression and weight gain but have no idea what to attribute that to, there’s a possible clue!) • LPS increases serum cholesterol levels by increasing hepatic cholesterol synthesis ( sidebar, that production is hepatoprotective from low-grade endotoxemia) • LPS influences metabolic endotoxemia, initiating obesity and insulin resistance • LPS causes an increase in intestinal tight junction permeability.
Speaking of Permeability, We Also Currently Understand That:
• Intestinal permeability and adipose tissue inflammation have long been considered mechanistic links in the relationship between diet, obesity, and chronic metabolic disease. • Intestinal permeability influences the translocation of LPS. • The translocation of LPS also influences adipokine secretion and adipocyte hypertrophy. • Gut permeability and LPS influence visceral fat mass and intra-abdominal fat mass. • Gut permeability increases systemic inflammation.
Speaking of Inflammation, We Also Currently Understand That:
• LPS influences inflammation, which in turn pings the positive-feedback loop for inflammation involving TRPV 1 pancreatic receptor. • Inflammation up-regulates the production of insulin (ps insulin is an anti-inflammatory hormone for all you insulin haters) • Inflammation up-regulates the production of cortisol. • Cortisol is a sterol, which is a subgroup of steroids that has oxygen at the end of the molecule. • Clinically significant gram-negative bacteria genera that produce LPS are all anaerobic. • Anaerobic bacteria cannot live or reproduce when oxygen is present. • Vitamin D, orange juice, and melatonin can cool LPS inflammation (this is an important clue).
Speaking of Anaerobic Bacteria That Cannot Live or Reproduce in the Presence of Oxygen, We Also Understand Thus Far That:
• LPS induces the production of reactive oxygen species. • Reactive oxygen species (ROS) are used by LPS-producing bacteria to suppress the growth of competitors. • The production of oxygen-based radicals, ROS, is the bane of all aerobic species. • The detoxification of reactive oxygen species is paramount to the survival of all aerobic life forms. • ROS have a role in cell signaling, including apoptosis and gene expression. • The accumulation of reactive oxygen species plays a role in many pathologies, especially neuropathologies. • There is a long-established connection between low vitamin D and a wide range of metabolic and inflammatory diseases. • Increasing Vitamin D intake can reduce inflammation in the body. • Vitamin D is made from cholesterol, and the activated form of Vitamin D is a sterol. • Sterols are a subgroup of steroids that have oxygen at the end of the molecule. • Orange juice consumption with a high-fat, high-carbohydrate meal prevents post-prandial increases in LPS concentration. • Orange juice contains terpenoids. • Terpinoids are a class of naturally occurring organic chemicals with oxygen-containing functional groups. • The role of the pineal gland’s main hormone, melatonin, plays a role in the regulation of ghrelin synthesis. • The surgical removal of the pineal gland (and subsequent production of melatonin) results in weight gain, fatigue, and sadness. • The role of melatonin also plays an important role in leptin and MC4R regulation. • Of the anorexigenic signals, leptin plays a vital role as a satiety molecule. • Melatonin significantly reduces food intake. • Melatonin also reduces hormone signaling from ghrelin, and neuropeptide Y (NPY)(aka the most potent central enhancer of appetite). • The long-term absence of melatonin leads to leptin resistance. • Melatonin decreases LPS-induced blood-brain-barrier disruption. • Melatonin, as well as its metabolites, is effective in scavenging a variety of ROS. • Melatonin also significantly increases oxygen saturation compared to basal measurements in trials (which is often seen as an effect with low clinical significance.) Question: is it possible that the mechanism that makes Vitamin D and Melatonin so important to our health is simply a function of increasing oxygen saturation, thereby making the environment more aerobic and thus less hospitable to LPS producing anaerobes while improving the environment for our symbiotic aerobic bacteria?
Full Circle
It is also well-known that the physiological control of appetite in animals is regulated by a complex interplay between hormones, neurotransmitters, and neuropeptides that interact reciprocally both at the central and peripheral level to stimulate or inhibit feeding behavior.
Perhaps we recognize that there is a microbial event influencing all of the outcomes mentioned above. Maybe therein lies a missing piece which made Stephan Guyenet and Gary Taubes both correct. Now it’s time to come full circle with it. Thus far, we also understand that:
• The microbiota is influenced by several factors including diet, use of antibiotics, hygiene, and genetics.
• Different populations of peoples have vastly different microbiotic profiles, and thus, may respond differently to different diets.
• Microbiomes influence the ‘healthy,’ or ‘unhealthy’ nature of foods on an individual basis (ahem, tomatoes).
• Gut dysbiosis and improving intestinal permeability are relevant targets for both the prevention and reversal of weight gain.
• People with weight loss resistance also tend to have a lower amount of Bacteroides, Verrucomicrobia, Faecalibacterium, and Prausnitzii and a higher amount of Actinobacteria and Firmicutes, and vice versa.
• The two most abundant bacterial phyla in humans are Firmicutes and Bacteriodetes.
• The ratio of Firmicutes to Bacteroides species will impact the efficiency of energy extraction, as well as changes in host metabolism, including absorbed calorie potential (hey-o)
• The Low-carbohydrate, ketogenic diet lowers Firmicutes and raises Bacteroides.
• A high-carbohydrate, high-fiber diet raises Firmicutes and lowers Bacteroides.
Next, let's discuss the DIETFITS trial.
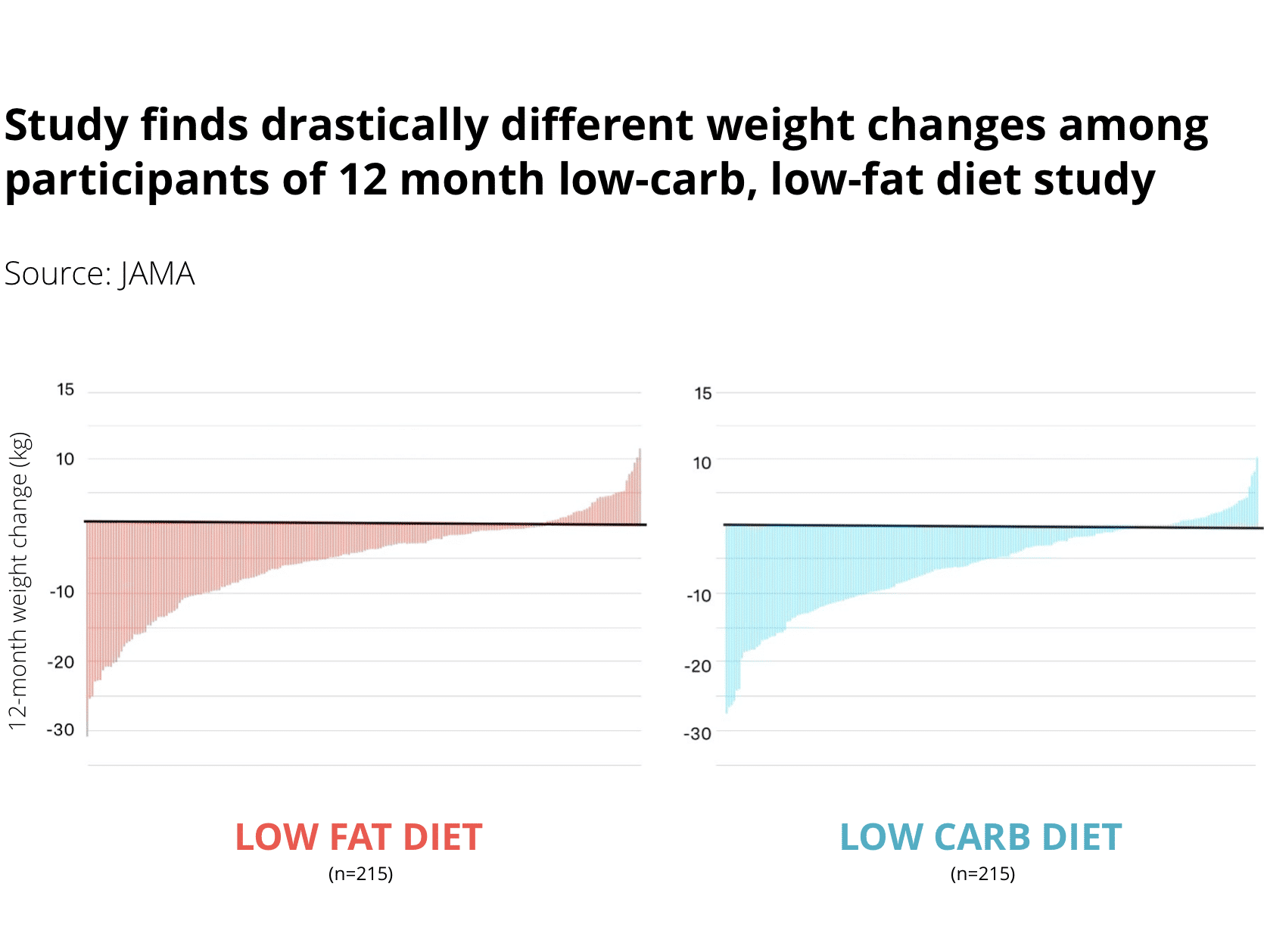 The year-long randomized clinical trial ( DIETFITS ) found that a low-fat, and low-carb diets produced similar weight loss and improvements in metabolic health markers — as a whole. The graphs, however, would tell a very different story if you looked at each line and each participant more carefully. Something very curious occurred, leaving us with a group of outliers for both low-fat and low-carb groups.
Now, as a whole both low-carb and low-fat groups showed similar outcomes. But on an individual level, it showed that while most would lose weight in both groups, there were some outliers who would gain a lot, too.
It is often said that any diet adhered to long enough will result in weight loss. Perhaps this is somewhat of a specious axiom in light of that trial and should be retired. Nevertheless, I have yet to see anyone else discuss those studies and ask the pertinent questions, like, "What could account for the reasons why one person gained weight, while the others lost weight?
Question: Is it possible that what causes one diet to benefit one and not another is directly related to the individual needs based on the microbiotic profile? Is it possible that the outliers in these groups that gained weight in the trial were selected to a group that further decreased Firmicutes or Bacteriodes in individuals who needed to increase those phyla, instead?
If Gary Taubes and Stephan Guyenet argued about the DIETFITS trial, Stephan would probably argue that there is no difference in the diets and talk about calorie-intake (missing the trees for the forest) while Gary would probably focus on the fact that some in the low-fat group gained weight, while the majority of the low-carb group lost weight (missing the forest for the trees). While that's just my speculation, the outcomes have always been fascinating to me.
Another point I’d like to make before my next point (and why I think the insulin-theory is terrible), the insulin production and tested genes had zero impact on predicting weight loss or weight gain outcomes. None. That is not intended as a jab per se to Taubes, I’d just like him to go back to the drawing board to reconcile that information.
In this next section, I'd like to address a few key points of contention that arose between Taubes and Stephan, using some helpful information about the gut microbiome. We understand that:
• The human large intestinal microbiota largely thrives on dietary carbohydrates originating from plant and fungal sources that are indigestible in the upper gut.
• Complex carbohydrates of high molecular weight (HMW) are required to nourish the microbiota of the large intestine. ( Get in-depth info on the importance of molecular weight in prebiotic fibers)
• For some low-carb proponents, despite their low-carbohydrate intake, many continue to gain weight. Perhaps they need to reduce calories (that’s a nod to CICO), or perhaps they needed to increase Firmicutes to improve homeostasis.
• Again, an improvement in the ratio of Firmicutes to Bacteroides species will impact the efficiency of energy extraction, as well as changes in host metabolism, including absorbed calorie potential.
• Some low-carb, high-fat proponents received the help of "safe-starch" promoter Paul Jaminet who showed that feeding gut flora with specific carbohydrates fostered metabolic improvements, and weight loss was experienced.
• Weight loss is associated with lowering the risk of all-cause mortality.
• Systematic reviews also reveal that low-carbohydrate intake is associated with an increased risk of all-cause mortality.
While Systematic reviews suggest that low-carbohydrate diets are associated with an increase in all-cause mortality, the RCTs, Systematic Reviews, and Meta-Analysis involving the full range of benefits associated with a low-carbohydrate ketogenic diet are incredibly compelling. (Here's the research review and summary on the outcomes of all available ketogenic diet RCTs published since 1920 ).
Therefore, there is more that remains to be teased out about low-carbohydrate diets, and to use the systematic reviews that suggest that low-carb diets are associated with an increase in all-cause mortality as a reason to dismiss low-carb or ketogenic diets altogether suggests dogmatic and dichotomous thinking.
Or perhaps sheer ignorance of the studies. That said, the ketogenic diet is not without adverse effects over time, which does indicate to the discerning mind that yet again, more nuance exists and deserves to be teased out accordingly:
• RCTs show that low-carbohydrate diets like the ketogenic diet may be beneficial for up to 8 weeks, however, this benefit diminishes over time, and significantly more adverse effects arise after 8 weeks.
• Perhaps low-carbohydrate and ketogenic diets increase Bacterioidetes and lower Firmicutes, but after 8 weeks of reduced fiber, Firmicutes becomes too low and must be increased for the purposes of maintaining homeostasis.
• Supplementation with specific types of fiber such as medium to HMW soluble carbohydrates, like hydrocolloids may prolong the benefits of a low-carbohydrate or ketogenic diet.
• When starved of complex carbohydrate substrate, the thick mucus that protects the intestines from pathogens can become a substrate for hungry bacteria.
• The hungry bacteria infect the colon wall and increase gut permeability.
• Increased gut permeability leads to the translocation of prolamins and glutelins to organ tissues.
• Low-carbohydrate diets, or simply diets devoid of complex carbohydrates (of varying MW) could be involved in the etiology of autoimmune disease, and other diseases related to increased intestinal permeability.
The year-long randomized clinical trial ( DIETFITS ) found that a low-fat, and low-carb diets produced similar weight loss and improvements in metabolic health markers — as a whole. The graphs, however, would tell a very different story if you looked at each line and each participant more carefully. Something very curious occurred, leaving us with a group of outliers for both low-fat and low-carb groups.
Now, as a whole both low-carb and low-fat groups showed similar outcomes. But on an individual level, it showed that while most would lose weight in both groups, there were some outliers who would gain a lot, too.
It is often said that any diet adhered to long enough will result in weight loss. Perhaps this is somewhat of a specious axiom in light of that trial and should be retired. Nevertheless, I have yet to see anyone else discuss those studies and ask the pertinent questions, like, "What could account for the reasons why one person gained weight, while the others lost weight?
Question: Is it possible that what causes one diet to benefit one and not another is directly related to the individual needs based on the microbiotic profile? Is it possible that the outliers in these groups that gained weight in the trial were selected to a group that further decreased Firmicutes or Bacteriodes in individuals who needed to increase those phyla, instead?
If Gary Taubes and Stephan Guyenet argued about the DIETFITS trial, Stephan would probably argue that there is no difference in the diets and talk about calorie-intake (missing the trees for the forest) while Gary would probably focus on the fact that some in the low-fat group gained weight, while the majority of the low-carb group lost weight (missing the forest for the trees). While that's just my speculation, the outcomes have always been fascinating to me.
Another point I’d like to make before my next point (and why I think the insulin-theory is terrible), the insulin production and tested genes had zero impact on predicting weight loss or weight gain outcomes. None. That is not intended as a jab per se to Taubes, I’d just like him to go back to the drawing board to reconcile that information.
In this next section, I'd like to address a few key points of contention that arose between Taubes and Stephan, using some helpful information about the gut microbiome. We understand that:
• The human large intestinal microbiota largely thrives on dietary carbohydrates originating from plant and fungal sources that are indigestible in the upper gut.
• Complex carbohydrates of high molecular weight (HMW) are required to nourish the microbiota of the large intestine. ( Get in-depth info on the importance of molecular weight in prebiotic fibers)
• For some low-carb proponents, despite their low-carbohydrate intake, many continue to gain weight. Perhaps they need to reduce calories (that’s a nod to CICO), or perhaps they needed to increase Firmicutes to improve homeostasis.
• Again, an improvement in the ratio of Firmicutes to Bacteroides species will impact the efficiency of energy extraction, as well as changes in host metabolism, including absorbed calorie potential.
• Some low-carb, high-fat proponents received the help of "safe-starch" promoter Paul Jaminet who showed that feeding gut flora with specific carbohydrates fostered metabolic improvements, and weight loss was experienced.
• Weight loss is associated with lowering the risk of all-cause mortality.
• Systematic reviews also reveal that low-carbohydrate intake is associated with an increased risk of all-cause mortality.
While Systematic reviews suggest that low-carbohydrate diets are associated with an increase in all-cause mortality, the RCTs, Systematic Reviews, and Meta-Analysis involving the full range of benefits associated with a low-carbohydrate ketogenic diet are incredibly compelling. (Here's the research review and summary on the outcomes of all available ketogenic diet RCTs published since 1920 ).
Therefore, there is more that remains to be teased out about low-carbohydrate diets, and to use the systematic reviews that suggest that low-carb diets are associated with an increase in all-cause mortality as a reason to dismiss low-carb or ketogenic diets altogether suggests dogmatic and dichotomous thinking.
Or perhaps sheer ignorance of the studies. That said, the ketogenic diet is not without adverse effects over time, which does indicate to the discerning mind that yet again, more nuance exists and deserves to be teased out accordingly:
• RCTs show that low-carbohydrate diets like the ketogenic diet may be beneficial for up to 8 weeks, however, this benefit diminishes over time, and significantly more adverse effects arise after 8 weeks.
• Perhaps low-carbohydrate and ketogenic diets increase Bacterioidetes and lower Firmicutes, but after 8 weeks of reduced fiber, Firmicutes becomes too low and must be increased for the purposes of maintaining homeostasis.
• Supplementation with specific types of fiber such as medium to HMW soluble carbohydrates, like hydrocolloids may prolong the benefits of a low-carbohydrate or ketogenic diet.
• When starved of complex carbohydrate substrate, the thick mucus that protects the intestines from pathogens can become a substrate for hungry bacteria.
• The hungry bacteria infect the colon wall and increase gut permeability.
• Increased gut permeability leads to the translocation of prolamins and glutelins to organ tissues.
• Low-carbohydrate diets, or simply diets devoid of complex carbohydrates (of varying MW) could be involved in the etiology of autoimmune disease, and other diseases related to increased intestinal permeability.
Speaking of Caloric Potential:
• LPS-induced septic and non-septic animals extract energy and calories differently. • LPS also promotes inflammation, which influences fat metabolism and intra-abdominal fat storage. • These points all support the notion that carbohydrates play a vital role in the production of LPS. • These points all support the idea that fat metabolism does not behave as expected with LPS-induced septic individuals. • A low Basal Metabolic Rate (BMR) is considered to be a factor predisposing an individual to obesity. • Low BMR is a predisposing factor for obesity, as well as increased insulin resistance (IR) • Mice injected with LPS experience reduction in BMR, and significant differences in metabolic rates corresponded with significant differences in cytokine levels. • PCOS is an LPS-mediated metabolic disease, showing an increase in cytokine production. • A study looking at BMR in different women found that women without polycystic ovarian syndrome (PCOS) needed 1553.87 ± 805.90 calories per day. • Women with PCOS have a lower BMR and needed only 1461.28 ± 75.72 calories per day. • However, women with PCOS who also had insulin resistance had an even lower BMR, needing only 1174.56 ± 454.38 calories per day. Although CICO still adds up in the above set of points, it should give us more pause by how we should approach the idea of “reduced calorie diets.” Perhaps practitioners (and obesity researchers alike) have not given this information its due consideration, or are simply unaware. Still, this is both a nod to CICO and a nod to what Gary was noting. The above-mentioned discussion about BMR should also lend itself to discussing hunter-gatherer populations like the Pima, as well as the high-carbohydrate Hazda. If high-carbohydrate consumption was altogether to blame for insulin resistance, inflammation, and obesity in the Pima, it should also be the same for the Hazda. However, this is not so. Here is some information about these two groups, and what we currently understand about the microbiome, to explain some of these discrepancies. • Gut Microbiome composition is associated with geographical proximity. • Upon adopting an agricultural diet, newly-minted agrarians experienced shorter stature, dental caries, cancer, inflammation, metabolic disorders, and other pathologies according to physical and forensic anthropologists. • Today, we see previously hunter-gatherer populations who recently switched to “Western Diet” get disproportionately higher rates of “Western Disease” compared with individuals in the same geographic region who have consumed a Western Diet for generations. This is especially true with type II diabetes, obesity, and autoimmune disease. • However, it is clear that today the post-agricultural diet has rendered many populations who are seemingly impervious to the scourges of their early-agrarian ancestors. • Microbiota evolves with their hosts over generations to the diet available. • Perhaps for a large population, perhaps the gut microbiota has evolved with hosts over time to withstand post-agricultural foods, including reduced complex carbohydrate intake. • However, perhaps for the Pima who were new to the “Western,” “post-agricultural diet” the microbes had not had enough time to evolve to withstand a diet that was as high in low-molecular-weight carbohydrates as it was low in high-molecular-weight, complex carbohydrates. • Thus, the diet disproportionately offered substrate to more proximal bacterial, while starving more distal bacteria that require more complexity. • Excessive consumption of low-molecular-weight, refined carbohydrates (LMW) is associated with an increase in LPS. • Conversely, increased consumption of complex carbohydrates is associated with a decrease in LPS. • Increased LPS, cytokine production, and insulin resistance are also associated with low BMR, as above described. • Is it possible that the Pima women did, in fact, have lower caloric intakes and experienced obesity? • E. Coli-induced sepsis and LPS injection into mice can cause significant differences in adiposity when compared to healthy mice fed equal calories.
Speaking of Molecular Weights, Complexity, and Carbohydrates:
• The Hazda consume a tremendous amount of fiber throughout the year from prebiotic-rich tubers and fruit from the baobab tree, something to the tune of about 100 to 150 grams of various molecular weights of fiber each day. • The Hazda have higher levels of Firmicutes and have higher levels of microbial richness and biodiversity. • The Hazda eat many carbohydrate-rich foods, including copious amounts of honey, yet do not experience the metabolic syndromes as the Pima. • The large intestine and especially lower colonic bacteria fed by high molecular weight carbohydrates exert disproportionate pressure on the rest of the gut microbiome by inducing changes to pH and producing higher amounts of short-chain fatty acids (SCFAs) acetate, butyrate, and propionate. • These SCFAs are preferred substrates for the epithelial cells of the large intestine. • The SCFAs produced by lower colonic bacteria prevent and repair intestinal perforations, thereby reducing permeability. • Is it possible that the regular consumption of high molecular weight complex carbohydrate intake protects against gut dysbiosis from a carb-rich and sugar-rich diet of honey, fruit, and tubers? • Is it possible that their BMR is higher than their Western counterparts? • Is it possible that this is why they are protected from obesity and metabolic derangements? Lots of questions. I know I said that I wanted to address these points very quickly, but I got carried away which is why I typically keep my mouth shut because that was a lot of typing. My point is, both Gary and Stephan have pertinent information. But instead of recognizing why each point was worth addressing, nothing was actually accomplished. Maybe next time someone will force them to work together to look for clues as to what could make sense of the outliers because they hold the key to forming a bigger, clearer picture. Why anyone would want to ignore the outliers is beyond me. Why anyone would want to ignore the majority and the overwhelming evidence spanning several decades is beyond me too. Now I understand, it’s personal there for them. And this makes for skyrocketing ratings and gossip like this was a tv novel. But do we really want to turn nutritional science into some dichotomaniacal pseudo-elitist circle-jerk? Can we not try to hold these two perspectives at face-value to see which warrants more inspection, and tease them out? And if we can’t set aside our dogma and our biases to at least look for more solid conclusions, that’s not science. That is a shitshow. Shouldn't we hold ourselves to higher standards than the Kardashians? Yours truly, Your Friendly Microbiome hobbyist and Microbiome diet enthusiast Karen
Related Reading
Share:



